 The membrane’s permeability depends on its structure: whether it has a system of pores (inorganic materials) or is a molecular sieve (organic materials). In the case of these devices, the membrane itself is the source of the main issues. The lower measuring range depends on the membrane’s permeability. These devices can be used to determine the pressure up to about 0.1 mmH 2O, which in practice means measuring polymer and biopolymer solutions of up to 2000 kDa. One of the chambers is filled with pure solvent, while the other with the studied solution and the difference in the hydrostatic pressure between the two chambers is measured. Because of that, the osmotic pressure is considered as one of the colligative processes.Ī membrane osmometer consists of two chambers divided by a membrane with specific pore sizes, which allow the solvent to move. If the osmotic equilibrium takes place in a solution-clear solvent system, the dilution of the solution becomes so significant that the dissolved substances presence becomes suppressed and as a result, the pressure of solvents vapours over the solution becomes equal to the vapour pressure over the clear solvent. From the solvents perspective, it is a natural intent to balance the chemical potentials, which results in the dilution of the solution with higher concentration. From the point of view of the molecules present in the solution, it is a phenomenon opposite to diffusion. The solvent moves through the membrane from the solution with lower concentration of the dissolved substance (or from the area of clear solvent) towards the solution with higher concentration. The osmosis process takes place between the solution and a clear solvent, or between solutions of different concentrations, provided they are separated by a membrane, which is permeable only to the solvents molecules ( Figure 2). The osmotic measurements can therefore be used to determine the molar mass, or for multi-molecular substances with significant polydispersion, to determine the average osmotic molecular mass. The difference of the vapour pressure would in this case be directly proportional to the molecular mass of a dissolved substance. If the non-volatile substance is a low-molecular-weight chemical compound, the changes in the vapour pressure can be explained by common phenomena, such as association or solvation, which are a result of interactions between molecules. Phase equilibrium for pure water and solution. The third virial coefficient provides information on mutual interactions between the biopolymer molecules, as well as its tendency to aggregate. The second virial coefficient obtained based on this relation allows characterisation of biopolymer-solvent interactions, and thus biopolymer tendency to solvation. Analysis and interpretation of osmometric research results is based on the van’t Hoff equation dependency of the concentration. The existence of the osmotic pressure, formed by diffusion of solvent molecules through a semi-permeable membrane, is the basis of this method. This method is successfully applied in the case of biopolymers that include polysaccharides. Membrane osmometry is one of the methods used in the studies of synthetic polymers to determine their average molecular mass and the degree of interaction between a polymer and a solvent. 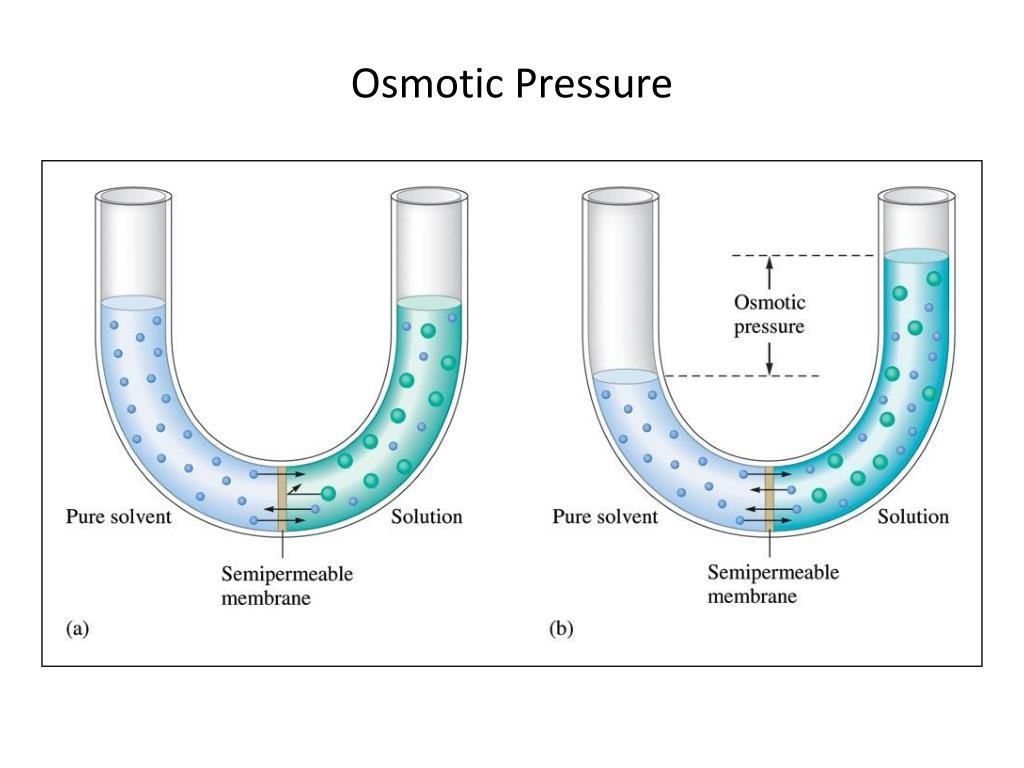 
Furthermore, an important issue is the effect of time on the osmotic properties of polysaccharides’, because with time, the aforementioned effects may change. The knowledge of osmotic properties of these systems provides the basis to appropriate use of polysaccharides having comply with the relevant technology functions, impart the appropriate texture and forming the sensory properties of the final product. 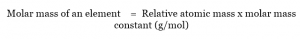
Osmotic properties of polysaccharides’ solutions and associated biopolymer-solvent and biopolymer-biopolymer type interaction are very important from a technological point of view. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
